More than 80 per cent of Australians are concerned about the prospect of side effects associated with vaccines against COVID-19, although more than half of Australians say they would definitely get a safe and effective vaccine if offered to them.
Subscribe now for unlimited access.
$0/
(min cost $0)
or signup to continue reading
A new study into attitudes towards vaccination shows an increase in the number of Australians who say they definitely or probably would get vaccinated from January this year, although there is more hesitancy than in August last year.
"When we asked a similar question in January, only 43.7 per cent of Australians said they would definitely get a safe vaccine. This jumped to 54.7 per cent in April," report co-author Professor Nicholas Biddle said.
"However, this number is still lower compared to August 2020 when we first asked the question, with 58.5 per cent of Australians saying they'd get a safe vaccine at that point in time.
"Even so, it would appear that willingness to receive a hypothetical safe and effective vaccine has stayed quite high."
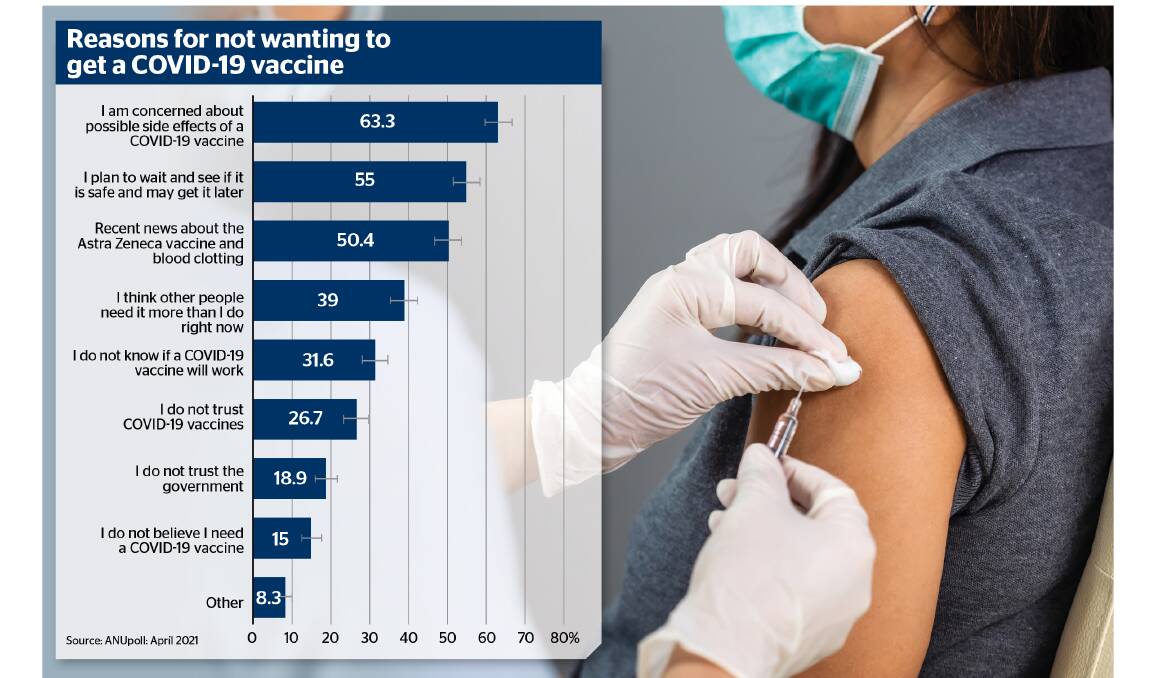
Almost 40 per cent of those surveyed by the Australian National University's Centre for Social Research and Methods said they were slightly concerned about side effects, while 20.3 per cent were moderately concerned and 21.8 per cent were very concerned.
Of those who said they probably or definitely wouldn't get the vaccine, 63 per cent said concerns about side effects were the main reason.
On top of that, 50.4 per cent of people who said they wouldn't take a vaccine said their decision was based on recent news about the AstraZeneca vaccine and blood clotting.
- Professor Nicholas Biddle
"On top of that, 50.4 per cent of people who said they wouldn't take a vaccine said their decision was based on recent news about the AstraZeneca vaccine and blood clotting," Professor Biddle said.
Government figures show the first day of stage 2a of the rollout, where the AstraZeneca vaccine has been made available to all Australians over the age of 50, hasn't led to a sharp increase in the number of vaccines administered. On Monday 56,364 vaccine doses were administered, but it's not the biggest day of the rollout so far.
READ MORE:
Up to 15.8 million doses are expected to be administered as part of the phase, which was brought forward after the Therapeutic Goods Administration advised the AstraZeneca jab shouldn't be used for people under the age of 50 because of the higher risk of developing blood clots in younger people.
It's expected the administration of the vaccines will ramp up after May 17, when GPs will be able to book over 50s in for their vaccinations.
States and territories have reported high numbers of new bookings to get the jab since the new phase opened this week.
Our journalists work hard to provide local, up-to-date news to the community. This is how you can continue to access our trusted content:
- Bookmark canberratimes.com.au
- Download our app
- Make sure you are signed up for our breaking and regular headlines newsletters
- Follow us on Twitter
- Follow us on Instagram


